A new Covid vaccine has been approved for use. I’d like to provide some information on it.
First, I’m a supporter of vaccines, and was quite willing to take the mRNA vaccine. I took the initial Moderna 2 dose, followed by the first Moderna booster when it was available for my risk category. I then took the second booster, but decided to switch over the Pfizer’s mRNA, as I had seen some interesting medical data suggesting crossing over.
To emphasize the value of our Covid vaccinations, I provide you with this comment from a world known medical researcher and epidemiologist. Follow this expert.
“The Covid-19 vaccines are some of the most incredible, lifesaving medical interventions of the last century.”

There is a great deal of misinformation that has been irresponsibly posted by non-experts. I will post what I find to be factual information around this vaccine so that those who are hesitant with an mRNA platform have this excellent option. “The Novavax vaccine [was built with] a much older technology for vaccine development where, instead of injecting the genetic recipe, we actually inject the protein. It [uses] a combination of spike proteins that form what are called nanoparticles, which group together. The Novavax vaccine also has an adjuvant, an immune stimulant to get a better immune response. In some ways, this is an older technology. The protein is made outside of the human body and then injected into us, and that induces the immune response.” https://publichealth.jhu.edu/2022/what-you-need-to-know-about-the-novavax-protein-based-vaccine
The FDA authorized the EUA for Novavax, Adjuvanted on July 13, 2022 to “prevent Covid-19, including the most severe outcomes that can occur such as hospitalizations and death.”
Novavax, Covid-19 Vaccine, adjuvanted is a two dose primary series, 3 weeks apart. The vaccine contains the spike protein and Matrix-M adjuvant.
The spike protein is produced in insect cells. Therefore, people who had any issue with fetal cell line use during any process of the discovery or manufacturing can rest assured there is no fetal cell line involved in this vaccine. The FDA press release:
Let’s discuss the clinical trial data (US and Mexico). Here is the link for the study publication: https://www.nejm.org/doi/pdf/10.1056/NEJMoa2116185?articleTools=true
The trial was a Phase 3, randomized, observer-blinded, placebo-controlled trial in the US and Mexico, evaluating the efficacy and safety of the vaccine. The primary objective (or primary endpoint) was to determine vaccine efficacy against PCR-confirmed Covid (PCR still being the gold standard and extremely reliable) occurring at least 7 days after the 2nd dose. Vaccine efficacy against moderate-to-severe disease and against different variants were also assessed.
Key points from study:
Close to 30,000 participants in a 2/1 ratio (vaccine vs. placebo)
Any cases reported in the vaccine cohort were mild in severity, with only 114 moderate-to-severe and 4 severe.
Vaccine efficacy against severe Covid was 100%.
Vaccine efficacy meeting the definition of the primary endpoint among participants who were at high risk for acquisition or complications of Covid 19 was 91.0%
Whole-genome sequencing was done to determine the VOC. Alpha was the predominant variant (VOC).
The vaccine was safe with generally mild headache, myalgia, fatigue and malaise. No anaphylaxis, no evidence of vaccine-associated enhanced Covid-19, no events that triggered prespecified pause rules. No imbalance in myocarditis or pericarditis or vaccine-induced immune thromboses with thrombocytopenia.
Limitations of study:
Because prior vaccines had become available, it was more difficult to enroll older people, preventing a statistically significant efficacy in older adults.
Imbalance in number of unblinding requests by trial participants at early stages of the trial. Other vaccines had become available. This led to many in the placebo group to be allowed to elect out in order to receive a different vaccine, already under EUA.
Efficacy is limited to 3 months, but is consistent with the limited vaccine efficacy follow-up for EUA.
This trial involved VOC Alpha primarily. This vaccine does not yet have data against delta and Omicron variant.
All trial participants were allowed a cross-over (blinded) after the first series was completed 3-4 months post 2nd dose. This has been done consistently in all the Covid trials, as it was felt that it was unethical to not allow placebo participants to not be properly vaccinated post initial 3 months of trial, if they chose. (Keep this thought in mind. *)
Therefore the durability of vaccine efficacy after the cross over may be less reliable.
*(As an aside for educational purposes: I’ve seen an absurd comment by a self-proclaimed “expert”, that the FDA lost the data in the pediatric approval of an mRNA vaccine. This expert clearly does not understand. No data was lost. The placebo group was lost to follow-up because they were given the option to receive the actual vaccine. Therefore, the placebo group no longer exists – i.e., there is a lost placebo group. Again, data is not lost. And no, the FDA didn’t lose any data, and no the FDA does not just go back to the company to get the data. Same situation here with Novavax allowing cross over for ethical reasons. Please, folks, check the “expert” you choose to follow. Find someone who knows something about clinical trial design, double blinded, cross-over analysis, etc.)
Said “expert” gets his medical information from the Gateway Pundit. Please folks, medical people do NOT use pundits from ANY media to access accurate medical information.
His lack of expertise exposed:
Never in 40 years of pharma/biotech work have I ever seen the FDA or pharma/Biotech “lose” the data. His comment is absurd and obviously conspiracy theory.
This expert even goes further to indicate how we can find out who “lost’
the data!!!! There is no data loss, as I have explained above.
Folks, be careful of the people you are getting information. There is too much disinformation put out by people who make wild conspiracy theory claims. Don’t buy into their garbage.
But it was good for a laugh around the table of researchers.
Finally to Novavax information:
Here is the full disclosure for patients and caregivers: https://www.fda.gov/media/159898/download
Another big blunder some “experts” are trying to tell you – that the vaccine is not necessary because we are done with the only surge, blah, blah, blah. There is no truth to only one surge per pandemic. This is one example of someone who doesn’t understand virology. The red “FALSE!!” and arrow are my addition in the memo below.
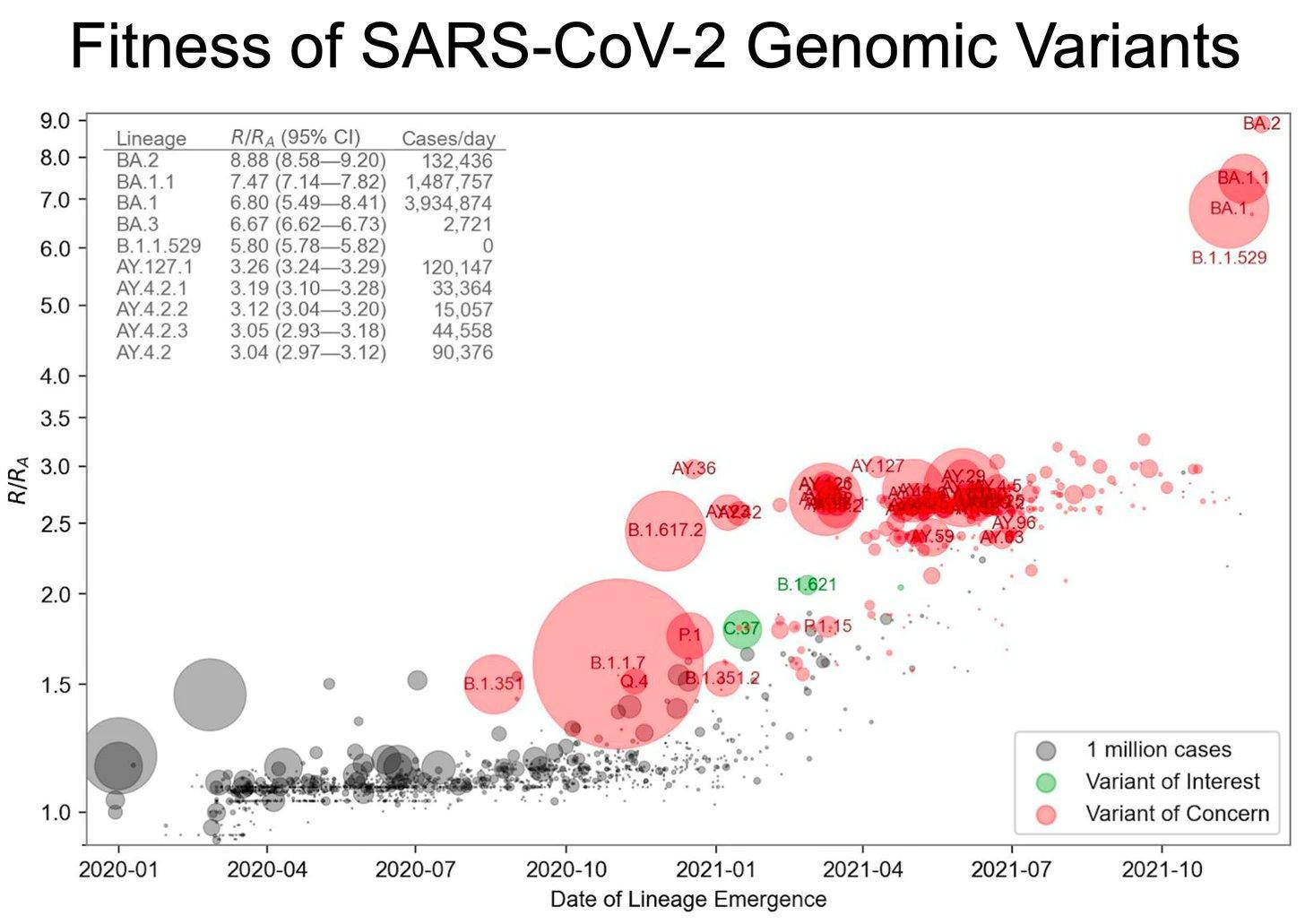
In contrast, this is what our scientists look at in graphing a pictorial of the VOC across time (we do NGS Next Generation Sequencing):
I can assure you that our NGS is sequencing Sars-CoV-2, and not any other pathogen. Covid is still here!
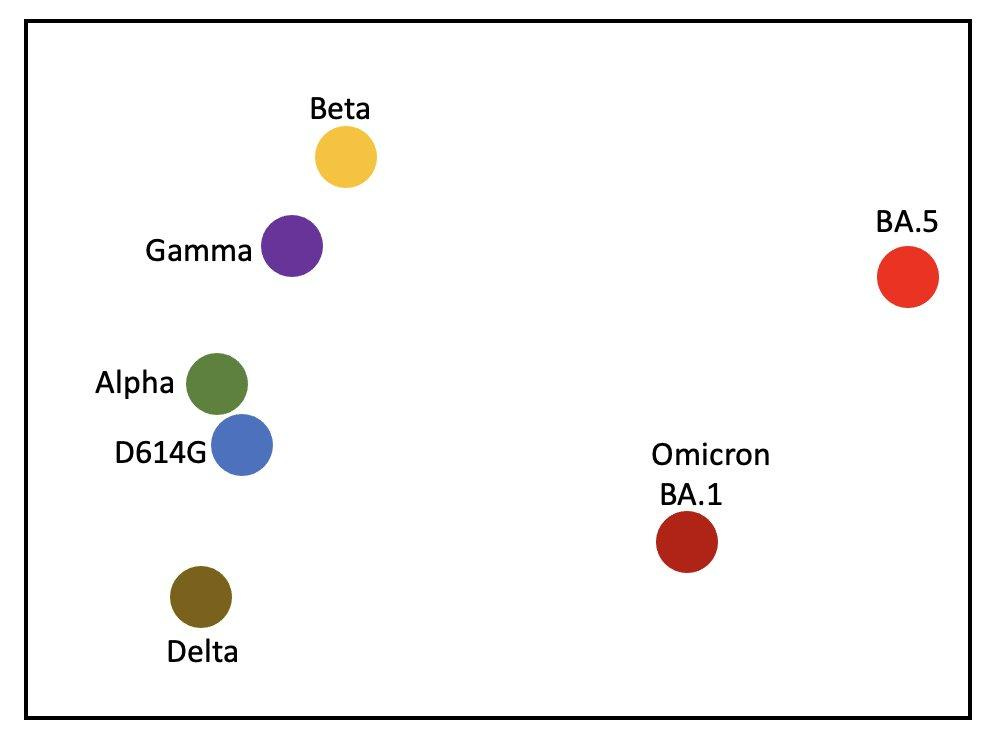
Here is a simple version for us:
Covid is still here. Do NOT let anyone tell you otherwise.
As my final aside – another reason to consider vaccination. It is now appearing that reinfections, no matter how mild the disease, may be contributing to Long Covid health issues. No one wants to bargain with that – Long Covid is a life changer. . . and not in a good way.
We do still need to immunize. Contrary to what some might say, we are NOT done with Covid. . . .or rather this virus is not done with us. Getting vaccinated now, either with boosters (if indicated) or the initial schedule will increase our protection individually and as a community.
Talk with your doc again about getting vaccinated.